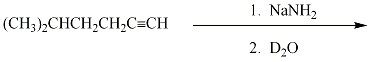Provide the major organic product for each of the following transformations.
The following solution is suggested to handle the subject “Provide the major organic product for each of the following transformations.“. Let’s keep an eye on the content below!
Question “Provide the major organic product for each of the following transformations.”
Answer
Carbonyl compounds are easily subject to a nucleophilic reaction, which involves the breaking down of carbon-oxygen \left( {{\rm{C = O}}} \right)
Below is the reaction of carbonyl compounds
Wittig reaction. This is used for organic synthesis to prepare alkenes using carbonyl compounds. After the reaction of carbonyl compounds with phosphorusylides, (2+2) is a cycloaddition reaction that forms a four-membered cyclic intermediary (oxophosphetane). This is followed by the formation phosphine dioxide to produce alkenes.
Below is the reaction scheme:

Formation of cyanohydrin: Carbonyl compounds in reaction with hydrogencyanide \left( {{\rm{HCN}}} \right)
Below is the reaction scheme:

Reaction of amine derivatives and carbonyl compounds
To make imine derivatives, the carbonyl compounds react with amines.
Below is the reaction scheme:

Carbonyl reduction: The reaction of carbonyl with Lithium aluminium hydroide \left( {{\rm{LiAl}}{{\rm{H}}_4}} \right)
Below is the reaction scheme:

Protecting carbonyl compounds

The reaction of cyclohexanone to $math_tag_1

Below is the mechanism of reaction of cyclohexanone and ethylene glycol

Below is the mechanism of reaction of cyclohexanone and phosphorus ylides

Below is the mechanism of cyclohexanone’s reaction to pyrrolidine

Below is the product to react cyclohexanone and aniline

Below is the mechanism to reduce cyclohexanone

Ans:
Below is the major organic product required for each transformation.

Conclusion
Above is the solution for “Provide the major organic product for each of the following transformations.“. We hope that you find a good answer and gain the knowledge about this topic of science.